J-Brain Cargo®
What is J-Brain Cargo®
The brain is protected by the blood–brain barrier (BBB), which also prevents many medicines from reaching the central nervous system (CNS). J-Brain Cargo® is JCR’s proprietary BBB penetration technology that enables drug delivery to the brain and spinal cord by targeting transferrin receptors expressed on brain vascular endothelial cells.

The Potential of J-Brain Cargo®
Advancing Treatment for Lysosomal Storage Disorders (LSD)

LSD are rare diseases caused by the deficiency or dysfunction of specific enzymes, many of which involve severe CNS manifestations. JCR applied J-Brain Cargo® to enzyme replacement therapy for mucopolysaccharidosis type II (MPS II) and obtained manufacturing and marketing approval in Japan. This marked the world’s first clinical application of BBB penetration technology in humans. Today, JCR is advancing research and development programs applying J-Brain Cargo® to more than 17 types of LSD.
Across Therapeutic Modalities
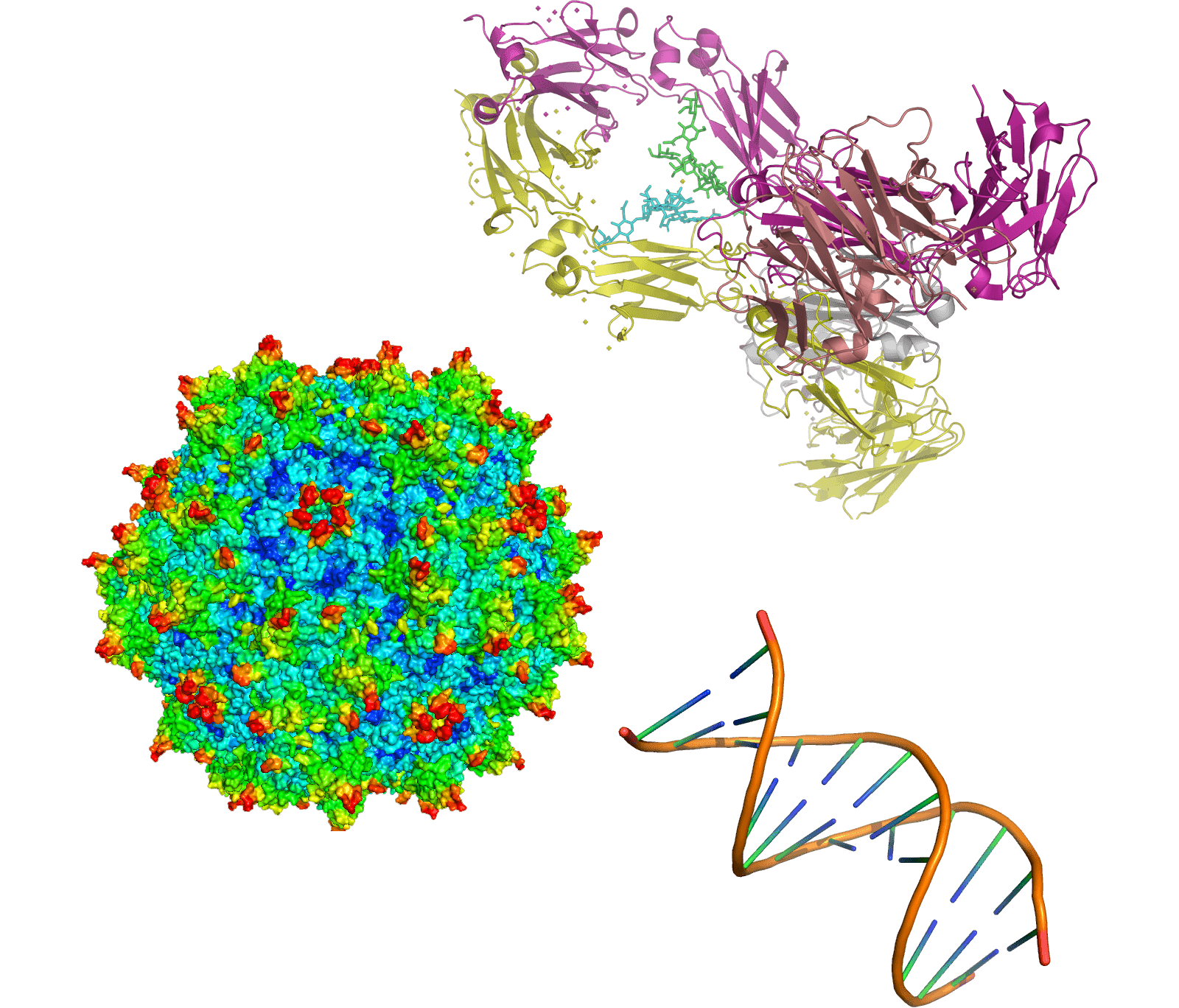
J-Brain Cargo® can be applied across diverse therapeutic modalities, including proteins such as enzymes and antibodies, oligonucleotides, lipid nanoparticles, as well as gene and cell therapies. It has the potential to enable delivery of therapeutics that cannot reach the brain alone or to further enhance their delivery.
Expanding Beyond Rare Diseases

"JCR first brought J-Brain Cargo® into clinical use in the rare disease field of LSD and continues to advance clinical development programs for multiple LSD indications. As a platform technology adaptable to diverse therapeutic modalities, J-Brain Cargo® also holds promise beyond rare diseases, including neurodegenerative conditions such as Alzheimer’s and Parkinson’s disease, as well as neuroinflammatory disorders and CNS tumors. JCR is actively pursuing collaborations with partners that bring cutting-edge expertise in each of these areas. "