Mid-Term Plan
JCR Pharmaceuticals Co., Ltd. (“JCR”) announced that JCR has formulated the Midterm Business Plan for FY2023-FY2027, “Reach Beyond, Together,” and hereby provides an overview.
Since our founding in 1975, we have pursued our business activities through proprietary research and development and a strong commitment to manufacturing, delivering to patients innovative medicines with world‑first mechanisms of action, including TEMCELL™ and IZCARGO™.
JCR is now one of the few companies in Japan that can operate the entire process of biopharmaceuticals, from research through to manufacturing. Having built foundational “R&D” and “Manufacturing” capabilities, we will continue to deliver value only JCR can provide to patients with rare diseases and their families.
Our Credo
Keyword
Reach Beyond, Together
<Positioning of this Plan>
Under the newly formulated Midterm Business Plan for FY2023-FY2027 “Reach Beyond, Together”, JCR will further augment the strengths we have developed through Midterm Business Plan for FY2015-FY2019 “HIYAKU” (Leap Into the Future) and Midterm Business Plan for FY2020-FY2022 “REVOLUTION” and create medicines that can only be made by JCR through its innovative drug creation platform technologies, making it a research-oriented specialty pharma with global exposure.
The keyword, “Reach Beyond, Together” express the unity of “Team JCR” as it moves toward a common goal, as well as the platform technology J-Brain Cargo® penetrating the blood-brain barrier and making progress toward new hope together with patients and their families.
Our Credo
Having built foundational “R&D” and “Manufacturing” capabilities, we can deliver value only JCR can provide to patients with rare diseases and their families.
Five Initiatives
[1] Creation of innovative core technologies
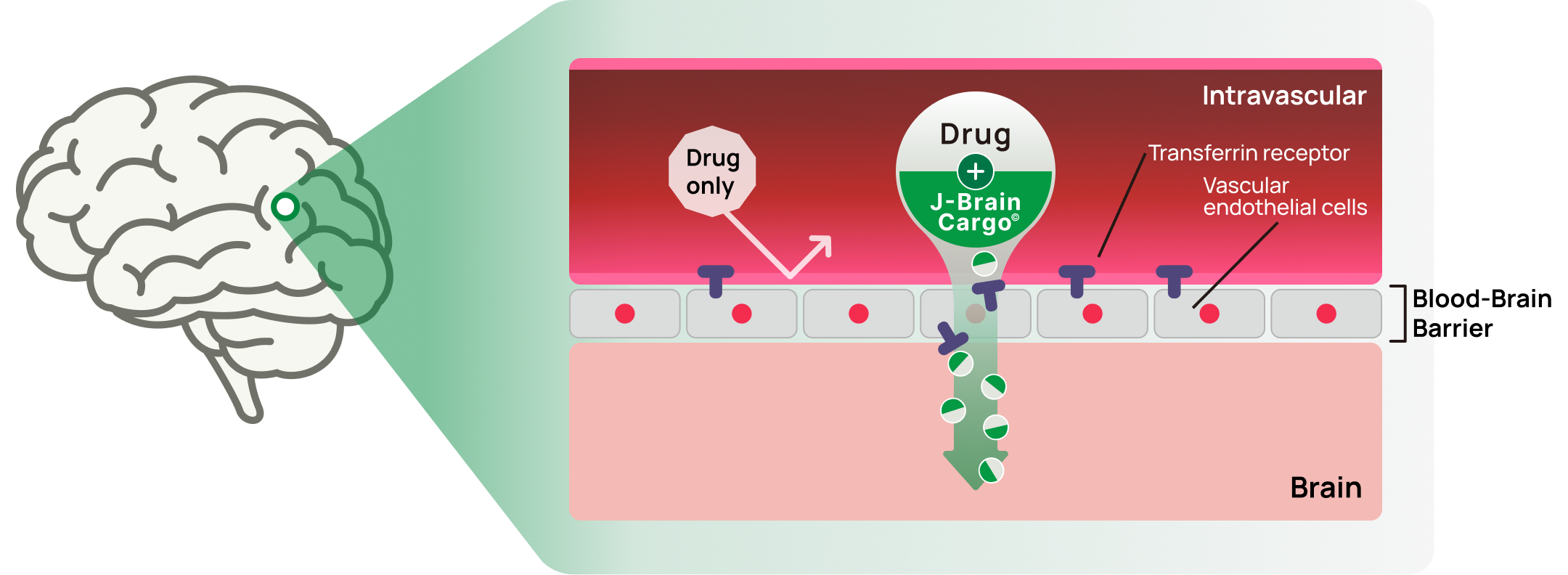
JCR will aim to create breakthrough therapies not limited to lysosomal storage disorders (LSDs) using a basic concept of J-Brain Cargo®, a platform technology JCR developed that was demonstrated as the first to penetrate the human blood-brain barrier.
Furthermore, to accelerate the development of next-generation J‑Brain Cargo® technologies capable of delivering therapeutic agents to tissues that have been difficult to access with conventional modalities—such as the eye, skeletal muscle, and cartilage—we are strengthening research activities through strategic collaborations with partners possessing world-class technologies and expertise in each respective field. Through these initiatives, we aim to expand our technology platform and enhance long-term corporate value.
About J-Brain Cargo®
J-Brain Cargo® is JCR Pharmaceuticals’ proprietary blood–brain barrier technology that enables the delivery of biopharmaceuticals to the central nervous system.
[2] Demonstrating of global standard production capacity
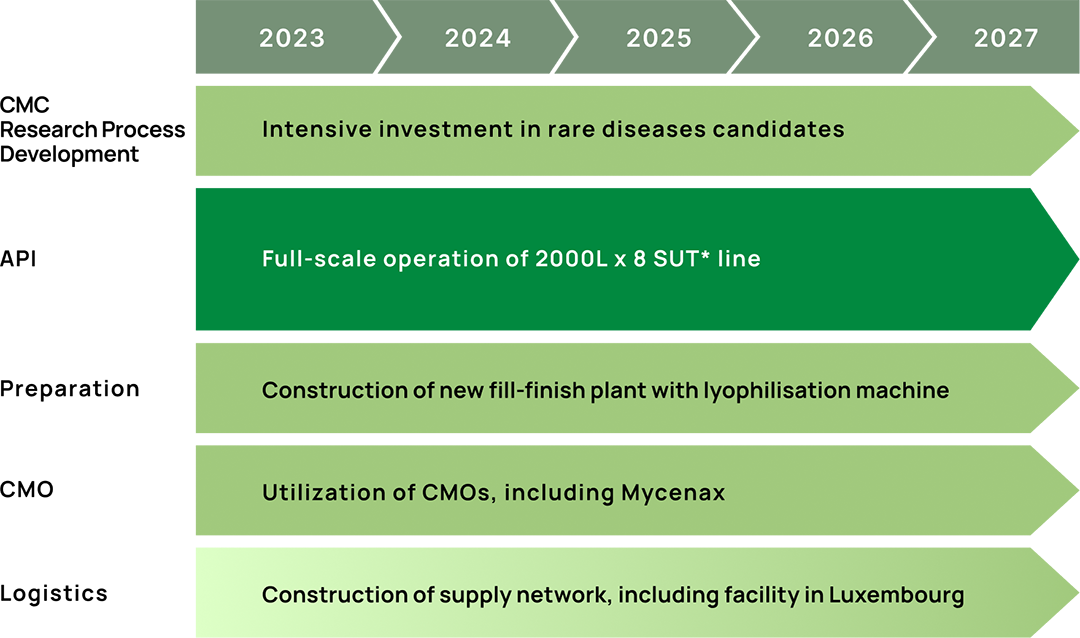
Our Strength in “MONOZUKURI (Manufacturing)”
Rooted in our founding commitment to excellence in manufacturing, we continue to advance our
capabilities in vaccine production. Our five production sites operate at full capacity, with close
collaboration across integrated research and manufacturing hubs in Kobe. We are building a resilient
global supply chain to respond to crises such as pandemics and natural disasters
Under our
Mid-Term Business Plan, we are investing around 40 billion yen in facilities and CMOs to boost our
manufacturing capabilities. We ensure a stable supply of growth hormone therapies, biosimilars, and
TEMCELL™ HS Inj. through integrated quality management. Leveraging our technologies and
expertise, we continue to take on new challenges in uncharted territory.
[3] Expansion of global quality assurance system in terms of quality and quantity
Expanding Our Global Supply
By establishing a quality-first culture and fostering talent with strong technical expertise, we are building a sustainable production framework.。
[4] Early launch of products for rare diseases
Leveraging its proprietary blood–brain barrier (BBB) crossing technology, J‑Brain Cargo®, JCR is advancing more than 17 drug development programs in the field of lysosomal diseases, including JR‑141 and JR‑171, both of which are currently undergoing global clinical trials. While many of these programs are still at the basic research or preclinical stages, they represent a robust and expanding pipeline. Among these candidates, JR‑471 targets fucosidosis, and JR‑479 is being developed for Tay–Sachs disease and Sandhoff disease—ultra‑rare disorders with extremely limited patient populations worldwide. JCR considers it its mission to deliver medicines that only JCR can create for such diseases that are difficult for large pharmaceutical companies to address. Through collaborations with strategic partners, we aim to achieve global commercialization of these innovative therapies.
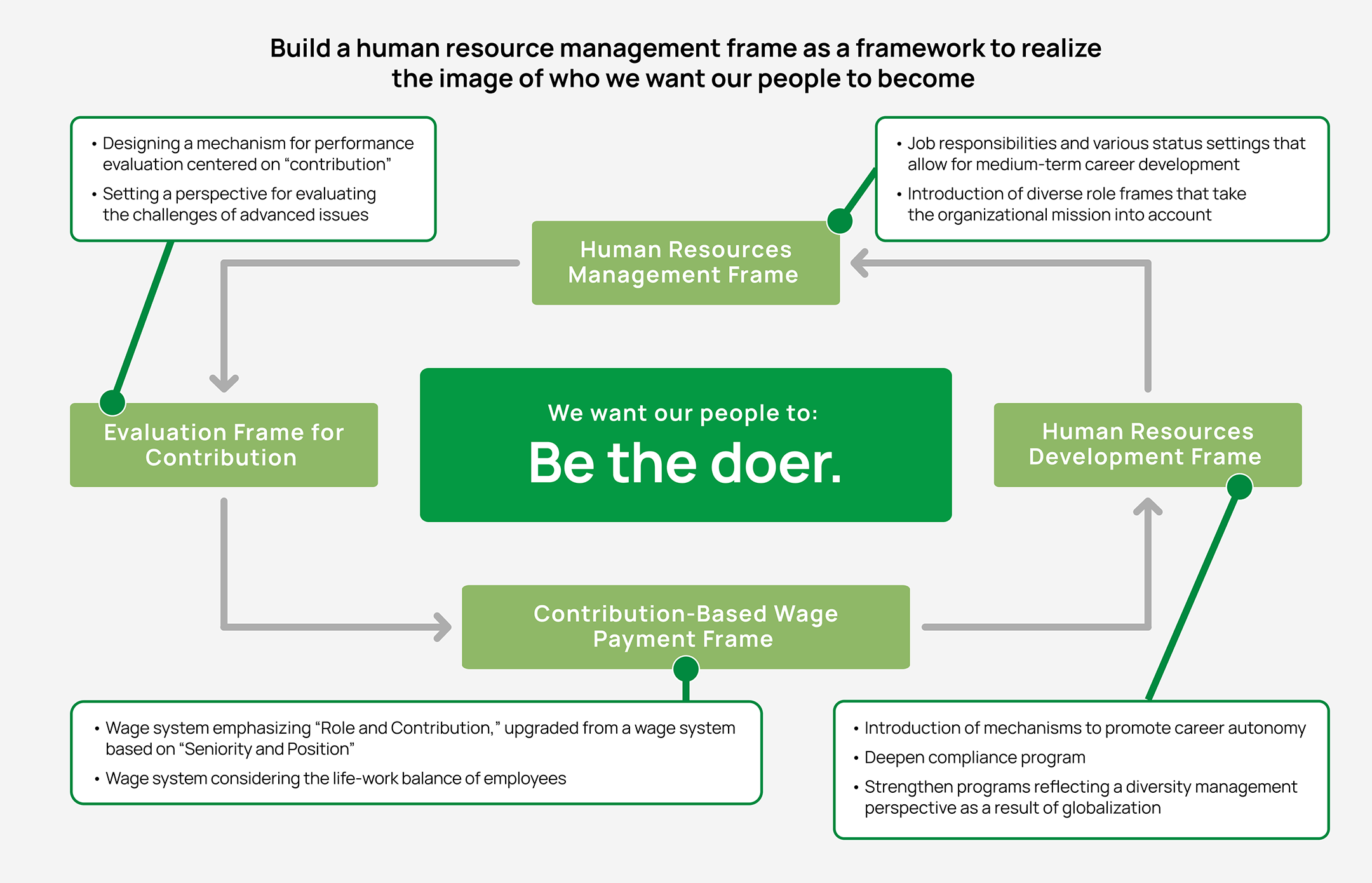
[5] Human resource development to support growth
JCR continues investment in human capital, and establish a human resource strategy that helps to improve corporate value. JCR is also building a human resource management frame as a framework to realize the image of who we want our people to become.
Building a dynamic talent portfolio that supports the execution of our strategy
- Assessment of the current talent portfolio
- Ensuring diversity through mid‑career hiring and creating synergies with existing talent
- Building an optimal talent portfolio
Promoting diversity and inclusion and embedding them throughout the organization
- Deepening R&D and manufacturing through the inclusion of diverse professional talent, leveraging their knowledge and experience
- Management and monitoring of inclusion
Driving the activation of individuals and the organization, and promoting higher employee engagement
- Strengthening engagement through clear communication of our vision
- Boosting organizational energy by supporting employee development and improving the workplace environment
Sustainability
JCR will achieve of sustainability through business activities based on the areas of Rare Diseases, Environment, Society and Corporate Governance.
Rare Diseases:
Rare diseases, in particular, are closely related to JCR's business activities and are a domain to which JCR can especially contribute.
We will proactively engage in things that only JCR can provide even for ultra-rare diseases where there are only few patients as we aim to achieve a society where no one will be left behind.
- Development of drugs for ultra-rare diseases and construction of new business models
- Efforts to improve access to medicines (Named Patient Supply) to deliver innovative medicines to people around the world
Environment:
- Investigation and implementation of measures to reduce environmental impact through business activities
- Promote environmentally friendly procurement on the premise of ensuring the quality and stable supply of pharmaceuticals
Society:
- Address the challenge of unmet medical needs by creating platform technologies
- Develop human resources so that each member of “Team JCR” can shine in their own station
Corporate Governance:
- Pursue governance in line with the company’s growth
- Ensure thorough compliance and strengthen risk management
